The First Law of Thermodynamics is a fundamental principle that connects energy, work, and heat. In the context of aircraft propulsion, it helps us understand how energy is transferred and transformed in engines such as jet engines, turboprops, and turbofans.
1. Understanding Energy in Thermodynamics
Energy can exist in various forms such as internal energy, kinetic energy, and potential energy. In propulsion systems, we are mainly interested in how energy changes during different thermodynamic processes inside the engine.
- Internal energy refers to the microscopic energy stored within the molecules of a substance.
- Heat is energy transferred due to temperature difference.
- Work is energy transferred when a force moves an object.
2. Statement of the First Law
The First Law of Thermodynamics states that:
Energy can neither be created nor destroyed, only transformed from one form to another.
For a closed system (no mass enters or leaves), the mathematical form is:
![]()
Where:
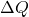 is the heat added to the system
is the heat added to the system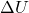 is the change in internal energy
is the change in internal energy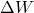 is the work done by the system
is the work done by the system
This equation is also known as the energy balance equation.
3. Application to a Control Volume
In aircraft propulsion, we often work with open systems (also called control volumes) where mass enters and exits, such as in a gas turbine engine. The First Law for a steady-flow process becomes:
![]()
Where:
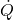 = rate of heat added
= rate of heat added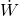 = rate of work done by the system (shaft work)
= rate of work done by the system (shaft work) = mass flow rate
= mass flow rate = specific enthalpy
= specific enthalpy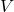 = velocity
= velocity = height
= height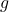 = acceleration due to gravity
= acceleration due to gravity
This equation considers both energy carried by mass (enthalpy, kinetic and potential) and energy added or removed as heat and work.
4. Relevance to Aircraft Engines
In a jet engine:
- Heat is added in the combustion chamber.
- Work is extracted by the turbine.
- Thrust is generated from the kinetic energy of the exhaust gases.
The First Law helps us analyze each component:
- Compressor: work is done on the fluid (negative work output)
- Combustor: heat is added
- Turbine: work is extracted from hot gases
- Nozzle: high-pressure gas expands and accelerates, increasing kinetic energy
By applying the First Law across these components, we can calculate engine efficiency, power output, and thrust generation.
5. Special Case: Isentropic Processes
If a process is adiabatic (no heat transfer) and reversible, it is called isentropic. In this case:
![]()
This idealization is useful in analyzing compressors, turbines, and nozzles where heat loss is minimal.

