Combustion is a chemical process in which a fuel reacts with an oxidizer, releasing heat and producing combustion products. In aircraft propulsion, combustion is at the heart of the engine’s energy conversion process, transforming chemical energy of fuel into thermal energy, which is then converted into mechanical work or thrust.
1. Basic Principle of Combustion
Combustion is typically a rapid oxidation reaction. For hydrocarbon fuels commonly used in aviation (like Jet-A), the simplified stoichiometric reaction with oxygen is:
![]()
This reaction releases large amounts of heat energy, which increases the temperature and pressure of the gases.
2. Stoichiometry
Stoichiometric combustion occurs when fuel and oxidizer are mixed in exact proportions needed for complete combustion, with no excess fuel or oxygen:
![]()
For a generic hydrocarbon fuel:
![]()
The air–fuel ratio (AFR) is the mass of air supplied per unit mass of fuel. In aviation gas turbines, combustion is typically lean, meaning excess air is supplied to ensure complete combustion and manage flame temperatures.
3. Heat of Combustion
The heat of combustion (or calorific value) is the amount of energy released when a unit mass of fuel is burned completely.
![]()
Aviation fuels have high energy density, making them suitable for propulsion. Typical Jet-A fuel has a lower heating value (LHV) of around 43 MJ/kg.
4. Combustion in Aircraft Engines
In gas turbine engines, combustion takes place in the combustion chamber (or combustor). The role of the combustor is to:
- Mix fuel and air thoroughly
- Sustain a stable flame
- Release energy at nearly constant pressure (idealized as an isobaric process)
- Minimize pressure losses
- Control exit temperature within turbine material limits
The combustion process can be idealized in thermodynamic cycle analysis as:
![]()
5. Combustion Process Modeling
The First Law of Thermodynamics for a control volume in steady-state combustion can be expressed as:
![]()
In practice, the heat added per unit mass of airflow is:
![]()
Where:
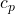 = specific heat at constant pressure
= specific heat at constant pressure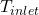 ,
, 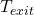 = inlet and exit temperatures
= inlet and exit temperatures
Combustion raises the temperature of the working fluid significantly, providing high-energy gases to drive the turbine.
6. Airflow Management in Combustors
Aircraft engine combustors use primary and secondary airflow paths:
- Primary zone: ~20–30% of air, supports stable combustion at stoichiometric or near-stoichiometric conditions.
- Secondary/dilution zones: Remaining air cools and mixes the products to achieve the desired exit temperature.
This staged airflow design:
- Prevents flameout at varying operating conditions
- Limits maximum gas temperatures to protect turbine blades
7. Emissions and Combustion Efficiency
Combustion efficiency is defined as:
![]()
Efficient combustors achieve >99% combustion efficiency. However, incomplete combustion can lead to:
- Unburned hydrocarbons (UHC)
- Carbon monoxide (CO)
- Soot (particulate matter)
Additionally, high flame temperatures promote NOx (nitrogen oxides) formation, which is a regulated pollutant.
8. Types of Aircraft Combustors
Several combustor designs are used in gas turbines:
- Can combustor: Individual flame tubes arranged around the engine.
- Annular combustor: Single continuous ring; compact and lightweight, common in modern engines.
- Can-annular combustor: Hybrid design with multiple cans arranged in an annular layout.
Design choice affects efficiency, emissions, stability, and maintenance.
9. Challenges in Combustion Design
Combustor design must balance:
- Complete combustion for high efficiency
- Flame stability across operating conditions
- Low emissions (NOx, CO, UHC)
- Minimal pressure loss
- Uniform temperature distribution at turbine inlet
Advanced designs use lean-premixed, rich-burn–quick-quench–lean-burn (RQL), and staged combustion concepts to meet these goals.
10. Role in Propulsion Cycle
Combustion is the energy-adding phase in the Brayton cycle:
- Compression increases pressure and temperature.
- Combustion adds heat at constant pressure.
- Expansion through turbines extracts work and accelerates exhaust gases for thrust.
Without efficient combustion, an engine cannot generate sufficient thrust or operate economically.
Understanding combustion is essential for analyzing and improving the performance, efficiency, and environmental impact of aircraft propulsion systems.

